 The numerator of each fraction is the number of ions that are a given distance away from the reference ion the denominator is the distance of the reference ion to the ions in question, as a multiple of the closest ( ie touching) internuclear distance between ions. The Madelung constant is the sum of all the ionic interactions in the crystal based on the three dimensional positions of the ions. Where A is the Madelung constant, ie a numerical factor which takes the relative positions of all the ions in the lattice structure into account in the calculation of the lattice energy. Neglecting the repulsive interaction at short interionic distances, we obtain: We can adapt the Coulomb expression to incorporate the ionic radii ( r + and r - for the cation and anion, respectively), the electron charge e, the magnitudes of the ion charges ( z + and z - for the cation and anion, respectively) and Avogadro's constant L. Where ε 0 is the permittivity of free space. The lattice energy of sodium chloride, for example, may be calculated to a reasonable approximation from first principles using Coulomb's law, which states that the electrostatic potential energy ( E) between two charged particles is proportional to the product of the charges( q 1 and q 2) and inversely proportional to the distance between them ( r): By comparing the values of lattice energy (directly or indirectly) through experiment provides a good test of the accuracy of the ionic bond model and thus a better insight into the nature of chemical bonding. We can challenge our students further by introducing them to the Madelung constant as a way of calculating the lattice energy of an ionic structure, given the atom positions in the crystal from X-ray diffraction studies. At A-level we build on this model and explain that an ion in sodium chloride has six neighbours and that the ions are arranged in a cubic lattice. We tell them that oppositely charged ions attract one another, and that the ions in the crystal lattice are, accordingly, ordered so that the immediate neighbours around a given ion have the opposite charge. When we teach ionic bonding at GCSE we usually show students the structure of sodium chloride. 
Saulsġ998.Sixthformers show off mathematical prowess in testing the ionic bond model Structure of Crystals Crystal Lattices Unit Cells From Unit Cell to Lattice From Lattice to Unit Cell Stoichiometry Packing & Geometry Simple Cubic Metals Close Packed Structures Body Centered Cubic Cesium Chloride SodiumĬhloride Rhenium Oxide Niobium Oxide Except as otherwise noted, all images, moviesīy Barbara L. Occupy the octahedral sites in the Na sublattice. The Na's occupy the octahedral sites in the Cl sublattice, and the Cl's 
We can look at the NaCl as made up of fcc / ccp lattices interpenetrating. Notice that there are 6 Cl surrounding the Na, and 6 Na around eachĬl. 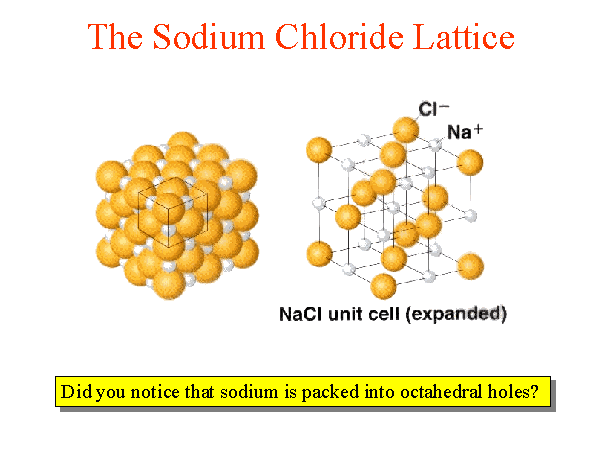
It may not be obvious, the red Cl's represent a different fcc / ccp unitĬell. There may be more than one way to divide a lattice into unit cells. The Cl sublattice must also be fcc / ccp. Since the repetition patterns of Na and Cl are the same in the lattice, If we compare this with the fcc / ccp unitĬell, it is clear that they are identical. If we take the NaCl unit cell and remove all the red Cl ions, we are Click on the image below to explore the open structure in The same structure, but with the ions moved further apart allows However, the tightly-packed structures make it difficult to These stack so: Click on the image below to view the NaCl lattice structure 
Click on the unit cell above to explore it in VRML. Sodium chloride also crystallizes in a cubic lattice, but withĪ different unit cell. Sodium Chloride crystal lattice SODIUM CHLORIDE 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
